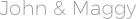I was going to try and put mine on XP with B.02 but I use the. also works but XP Ive heard conflicting reports about. I may send some old emails to you however, I am not sure if you can help. Have you bought this GC with software Is there a label with license number For Chemstation MSD B.02 is the last version that will operate a 5890. FDA wants to have the "metadata." And don't delete your data files you need to back up data and you need SOP covering that. Agilent G1701EA MSD Productivity ChemStation.
#Agilent chemstation software manual pdf#
In the view of FDA, if you can print various copies of the same chromatogram from Chemstation (or Class-VP, etc), the electronic record (the chromatogram) must comply with Part 11 requirements, even if you delete the electronic data after printing. OpenLab CDS ChemStation AIC M8304AA server pdf manual download. We have been requested to provide hard copies of audit trails as well. I have represented the lab to repond to FDA auditors when they asked to show the audit trails from the Dionex and Shimadzu LCs. Agilent FID Flame Ionization Detector user guide. Agilent Technologies Series Gas Chromatograph Service Manual. It can not generate audit trails with Chemstation version A, unlike many versions of Chromeleon (Dionex) or Class-VP (Shimadzu). Agilent Site Preparation and Installation Manual.
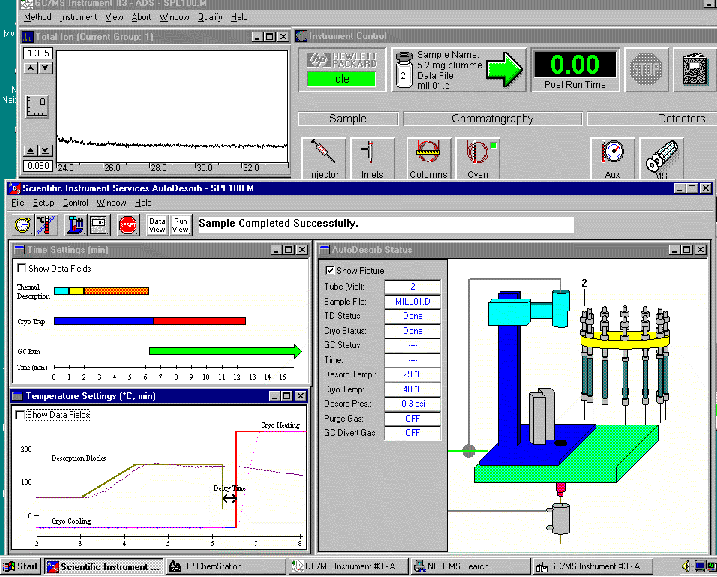
I am really sure that the ChemStation version A is not compliant and I have used it to run the Agilent LCs.